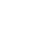Total hardness reagent plays a crucial role in water quality assessment across numerous industries and applications globally. Understanding water hardness – the concentration of dissolved calcium and magnesium – is paramount for optimizing processes, preventing scaling, and ensuring human health. The availability of accurate and reliable total hardness reagent is, therefore, a fundamental necessity for laboratories, treatment plants, and field testing operations worldwide.
The demand for effective water quality monitoring solutions is consistently rising, driven by stringent environmental regulations and increasing awareness of the impact of hard water on infrastructure and consumer products. Total hardness reagent, when manufactured to exacting standards, provides a cost-effective and efficient means of determining hardness levels, allowing for proactive management of water resources. This is particularly vital in regions facing water scarcity or where industrial processes heavily rely on specific water qualities.
Beyond industrial applications, accessible total hardness reagent empowers communities to test their water sources, safeguarding public health and ensuring access to safe drinking water. Its use extends from residential well testing to large-scale municipal water supply analysis, solidifying its importance as a cornerstone of water quality control.

The Significance of Total Hardness Reagent
The measurement of total hardness is fundamentally linked to the quality and usability of water. High hardness can lead to scale formation in pipes and appliances, reducing efficiency and increasing maintenance costs. Accurately quantifying total hardness using total hardness reagent allows for the implementation of water softening solutions, protecting infrastructure and extending equipment lifespan.
Beyond infrastructure concerns, hardness impacts various industrial processes, from boiler efficiency to the effectiveness of detergents and cleaning agents. Employing reliable total hardness reagent provides the data necessary to optimize these processes, reducing operational expenses and ensuring product quality.
Understanding Total Hardness Reagent
Total hardness reagent is a chemical mixture specifically formulated to react with calcium and magnesium ions present in water samples. This reaction produces a color change, or other measurable output, directly proportional to the concentration of these ions, enabling a precise determination of the water's total hardness. Commonly, EDTA (ethylenediaminetetraacetic acid) is the primary titrant used within these reagents, forming stable complexes with calcium and magnesium.
The underlying principle is a complexometric titration. The reagent’s ability to accurately bind to these ions is critical, and reagent quality dictates the precision of the measurement. Modern formulations often incorporate indicators to visually signal the endpoint of the titration process, streamlining the analytical procedure.
This technology directly supports modern industrial and humanitarian needs. Industries require accurate hardness measurements to prevent scaling and corrosion, while communities need it to ensure safe drinking water. Total hardness reagent serves as the linchpin in enabling these quality control processes.
Core Components of Total Hardness Reagent
The effectiveness of any total hardness reagent hinges on the purity and concentration of its key ingredients. EDTA, as mentioned, is the primary complexing agent. Its quality directly impacts the reagent's sensitivity and accuracy. Buffering agents are also crucial to maintain a stable pH during titration, ensuring consistent reaction kinetics.
Furthermore, indicator dyes play a pivotal role in visually determining the titration endpoint. Eriochrome Black T is a commonly used indicator, changing from wine red to a pure blue color when all calcium and magnesium ions are complexed. The sharpness of this color change is indicative of the reagent’s quality. Stabilizers are often added to prevent degradation of the EDTA or indicator over time, extending the reagent's shelf life.
Finally, the solvent used to dissolve these components is also significant. Distilled or deionized water is essential to avoid introducing impurities that could interfere with the titration process. Rigorous quality control during the manufacturing process is essential to ensure the consistent performance of each total hardness reagent batch.
Key Factors Influencing Reagent Performance
Several factors can significantly impact the accuracy and reliability of total hardness reagent. Temperature is a critical parameter, as EDTA complex formation is temperature-dependent. Maintaining a consistent temperature during titration, or applying appropriate corrections, is essential for obtaining accurate results.
The pH of the water sample also plays a crucial role. Optimal pH levels, typically around 10, are required for efficient complex formation. Buffering agents within the reagent help maintain this optimal pH range, but extreme pH values in the sample can still interfere with the titration.
Total Hardness Reagent Performance by Method
Global Applications of Total Hardness Reagent
The applications of total hardness reagent are far-reaching. In municipal water treatment plants, it's used to monitor raw water sources and assess the efficiency of softening processes. This ensures that treated water meets regulatory standards for hardness levels.
Industrial facilities, such as power plants and manufacturing companies, rely on total hardness reagent to prevent scale buildup in boilers, cooling towers, and other critical equipment, safeguarding operational efficiency and minimizing downtime. In post-disaster relief operations, portable testing kits containing this reagent are crucial for assessing the potability of water sources in affected areas.
Advantages and Long-Term Value
Utilizing high-quality total hardness reagent offers numerous tangible benefits. Accurate hardness measurements translate to cost savings by preventing scale buildup, optimizing chemical treatment processes, and extending the lifespan of equipment. Proactive water management contributes to environmental sustainability by reducing water waste and minimizing the need for harsh cleaning chemicals.
From a societal perspective, reliable water quality data enhances public health and builds trust in water providers. This peace of mind fosters a sense of security and well-being within communities. The inherent reliability of established methods, paired with the accessibility of total hardness reagent, makes it a crucial component of water safety programs worldwide.
Future Trends and Innovations in Reagent Technology
The future of total hardness reagent technology is focused on improved ease of use, increased accuracy, and enhanced sustainability. Development of pre-filled cartridges and automated titration systems are streamlining the analysis process, reducing the potential for human error.
Furthermore, research is underway to develop more environmentally friendly reagent formulations, utilizing biodegradable components and minimizing hazardous waste. Digital integration, with reagent bottles containing QR codes linked to online databases and calibration information, is also gaining traction.
The increasing adoption of Internet of Things (IoT) sensors and real-time monitoring systems will further enhance the value of total hardness reagent, enabling continuous water quality assessment and proactive management of water resources.
FAQS
The shelf life of total hardness reagent typically ranges from 12 to 24 months, depending on storage conditions and the specific formulation. It’s crucial to store the reagent in a cool, dry place away from direct sunlight and heat. Always check the expiration date on the bottle before use, as expired reagent can yield inaccurate results. Proper storage is essential for maintaining the reagent’s efficacy and ensuring reliable water hardness measurements.
Temperature significantly influences the complexation reaction between EDTA and calcium/magnesium ions. Higher temperatures generally increase reaction rates, but can also affect indicator sharpness. Most titration procedures recommend performing the analysis at a specified temperature (often 25°C). If the sample temperature differs, a correction factor may need to be applied to the reading to account for the temperature-induced error.
While most total hardness reagents are relatively safe, it's important to exercise standard laboratory safety precautions. Wear appropriate personal protective equipment (PPE), including gloves and eye protection. Avoid contact with skin and eyes. If contact occurs, rinse thoroughly with water. Refer to the Safety Data Sheet (SDS) for detailed information on hazards and handling procedures. Store reagent away from incompatible materials.
Total hardness represents the combined concentration of all polyvalent cations (primarily calcium and magnesium) in water. Carbonate hardness, on the other hand, refers specifically to the portion of total hardness caused by the bicarbonate and carbonate ions. Carbonate hardness can be removed by boiling, whereas non-carbonate hardness (the remainder) requires chemical treatment like softening. Understanding both is vital for comprehensive water quality assessment.
Regular calibration of your titration equipment is crucial for ensuring accurate results. The frequency of calibration depends on the equipment’s usage and manufacturer's recommendations, but a general guideline is to calibrate at least once a month, or after any maintenance or repairs. Use standardized solutions to verify the accuracy of your burettes and pipettes.
False positives can occur due to interference from other ions that may react with the indicator, or due to contamination of the sample. False negatives can result from expired reagent, incorrect pH levels, or insufficient reagent concentration. Ensure proper sample preparation, use fresh reagent, and maintain the correct pH to minimize errors. Follow the reagent’s instructions carefully.
Conclusion
Total hardness reagent remains an indispensable tool for water quality management across a diverse range of applications. From safeguarding public health to optimizing industrial processes, its ability to accurately measure calcium and magnesium concentrations is paramount. Continuous innovation in reagent technology, focusing on improved accuracy, ease of use, and sustainability, will further solidify its importance in the years to come.
Investing in high-quality total hardness reagent and adhering to best practices for its use is not merely a matter of compliance; it is a commitment to responsible resource management, environmental protection, and the well-being of communities worldwide. Visit our website at www.jensprima.com to explore our range of reagents and solutions for all your water quality testing needs.




















































































 Download
Download Video Center
Video Center Technical Services
Technical Services FAQS
FAQS Service Network
Service Network Privacy Policy
Privacy Policy Contact Us
Contact Us